Synthesis of Acetaldehyde and Paraldehyde[ Back to the Chemistry Archive ] ProcedureAcetaldehyde 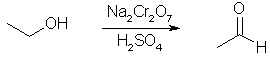
Place 260 mL of 23% sulfuric acid in a 1500-mL Florence flask. Connect an addition funnel to the flask, and set it up for vacuum distillation with a 500-mL receiving flask. The receiving flask should be immersed in a salt-ice bath to cool the distillate when it comes over. Although a setup for vacuum distillation is used, it is not necessary to apply a vacuum; the vacuum adapter merely provides a means of equalizing pressure. Prepare a solution of 100 g of sodium dichromate in 200 mL of water and 127 mL of ethyl alcohol. Factor in the amount of water that is already in the ethyl alcohol when making the solution, i.e. 100 mL of 95% alcohol has 5 mL of water in it already. Place this solution in the addition funnel, position the stem of the funnel so that it is about 3 cm above the surface of the acid. Heat the acid until it just begins to boil, then add the mixture in the funnel in a steady stream to the acid. It will not be necessary to heat the flask during the addition because it will generate its own heat. The heat will be sufficient to distill over the acetaldehyde along some alcohol and waste acetal. If acetaldehyde vapors begin to escape from the flask, regulate the distillation by decreasing the amount of dichromate solution being added. If the reaction flask does not boil on its own, gently heat it for a short time until boiling begins. Acetaldehyde Ammonia Trimer 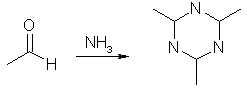
The crude acetaldehyde thus obtained is difficult to distill from the alcohol and acetal mixed with it. It is therefore converted to aldehyde-ammonia, then back to pure aldehyde. Place the crude acetaldehyde in a Florence flask of suitable volume to contain no more than two thirds of the liquid. Attach a reflux condenser to the flask. Fill the condenser jacket with 30 °C water. It is only necessary to have the warm water in the jacket, not flowing. Stop up the lower connecter to prevent water from leaking out. To the top of the condenser, attach a glass tube connected to a wash bottle filled with 50 mL of ether. This wash bottle is then connected to another wash bottle filled with 50 mL of ether. After all connections have been made, the crude aldehyde is heated to a gentle boil for 5-10 minutes. The ether will absorb the acetaldehyde as it boils off. If the ether begins to rise up into the connecting tube from the condenser to the wash bottle, increase the heating. Combine the ether into a 150-mL beaker immersed in a salt-ice bath. Bubble dry ammonia gas into the ether through a funnel or wide tube immersed near the bottom of the beaker. Add ammonia until the ethereal solution smells strongly of ammonia. After about an hour, pure ammonia-aldehyde should have separated out. Scrape the crystals from the beaker and collect them by suction filtration. Wash the crystals with a small amount of ether, and allow them to dry in a desiccator. The yield is about 30 g. To obtain pure acetaldehyde, dissolve 10 g of aldehyde-ammonia in 10 mL of water in a 50-mL Florence flask. Add 28 mL of cold 29% sulfuric acid to the flask and set it up for simple distillation. Heat the flask on a water bath to distill over the aldehyde. Place the receiving flask in a salt-ice bath to cool the volatile acetaldehyde. Paraldehyde (Acetaldehyde Trimer) 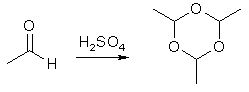
Acetaldehyde is extremely volatile and cannot be stored satisfactorily unless it is refrigerated or sealed in glass ampoules (not just capped), therefore it is necessary to prepare acetaldehyde each time it is needed. There is, however, an easier solution. A quick and easy way to store acetaldehyde is to polymerize it to paraldehyde, which can be handled and stored easily, then depolymerize when you need to use it. Now you can make a larger amount without worry. To polymerize acetaldehyde for storage, place it in a dry test tube and cautiously add 1 drop of concentrated sulfuric acid per 2 mL of acetaldehyde in the tube. Mix thoroughly, the polymerization will begin to take place. Some gentle warming can hasten the reaction. After some minutes add 3-4 mL of water per 2 mL of acetaldehyde, an insoluble precipitate of paraldehyde will form. As an aside, technically paraldehyde is a controlled substance. It is a sedative and a hypnotic drug useable by prescription only. To depolymerize paraldehyde back into acetaldehyde, place the paraldehyde into a round-bottom 200-mL Florence flask. Add 4-5 drops of concentrated sulfuric acid for every 20 g (20 mL) of paraldehyde in the flask. Set the flask up for fractional distillation, use glass in the fractionating column. Use a 125-mL Erlenmeyer flask as the receiver; keep it cool by immersing in an ice water (but not salt-ice) bath. Place a loose plug of cotton into the Erlenmeyer flask to help reduce evaporation loss; it must be loose. Care must be taken to prevent the cotton from coming into contact with the distillate. After setting up, heat the flask gently. The temperature of the distillate must not be allowed to rise above 35 °C as it will only repolymerize. The acetaldehyde is now ready for use. |