Introduction
Syringaldehyde has been prepared by the oxidation of eucalypt lignin with nitrobenzene and alkali. It has been methylated with dimethyl sulphate and mescaline has been synthesised from the 3,4,5-trimethoxybenzaldehyde so formed.
Mescaline, 3,4,5-trimethoxyphenethylamine (IV), the hallucinatory principle of "pellote", was isolated in 1896 by Heffter3, and its chemistry has been studied in detail. Numerous syntheses have been developed2 and many of these utilised 3,4,5-trimethoxybenzoic acid or one of its derivatives as starting material. Other synthetic routes made use of 3,4,5-trimethoxybenzaldehyde (II), and Slotta and Heller8 and Slotta7 prepared their starting material, trimethoxyphenylpropionic acid, by condensation of the substituted benzaldehyde with malonic acid and reduction of the resulting cinnamic acid. Mescaline was then obtained by Hofmann degradation of the trimethoxyphenylpropionamide.
Slotta and Szyska9,10 obtained mescaline directly by condensing II with nitromethane and reducing electrolytically the ω-nitro-trimethoxystyrene (V). The latter also has been reduced with lithium aluminium hydride5. Pure crystalline mescaline has also been synthesised by condensation of 3,4,5-trimethoxybenzaldehyde with potassium cyanide followed by acetylation and catalytic reduction to the amine4.
Syringaldehyde, 3,5-dimethoxy-4-hydroxybenzaldehyde (I), is an attractive alternative starting material. D.E. Bland and coworkers1 reported its preparation by the oxidation of certain eucalypt lignins with nitrobenzene and alkali. Working on a small scale they showed that Eucalyptus regnans (Mountain Ash), E. obliqiia (Messmate Stringybark) and E. diversicolor (Karri) gave better than 5% yields of syringaldehyde. It was decided, therefore, to study the preparation of mescaline from these three species.
The first stage is the methylation of the free phenolic group of syringaldehyde which can be accomplished either with dimethyl sulphate and alkali, or with diazomethane. Only the first method was examined in detail, since diazomethane would not be favoured as a large-scale reagent. Both the electrolytic and the lithium aluminium hydride reduction of V present difficulties on a large scale, especially in the decomposition of the lithium-alanate complex. On the other hand, cyanohydrin formation can be performed quite readily by the reaction of potassium cyanide with the aldehyde bisulphite compound, and catalytic reduction of III is straight-forward and cheaper. This route was therefore preferred.
Procedure
75 g portions of air-dry sawdust from kiln-dried timber were oxidised with nitrobenzene and alkali at 150°C according to the method of Bland1. The mean yield of syringaldehyde from E. regnans was 4.9%, from E. obliqua 3.1% and from E. diversicolor 3.2%. The use of a mechanical stirrer in the autoclave would probably have increased the yields, which may also depend on the particle size of the sawdust.
75 of sawdust gave the largest volume of solution which could be extracted conveniently by hand. For larger volumes, a continuous extractor was used in which hot benzene was allowed to flow through the aqueous phase, but during the long period in which the benzene extract was kept at 80°C, a large proportion of the he syringaldehyde decomposed. For large-scale Operation, the benzene extract would have to be kept at a lower temperature by the use of a climbing film or flash evaporator.
The crude extract which contained both syringaldehyde and vanillin was analysed using a mass spectrometer. since spectrophotometric analyses are of little use in distinguishing these two compounds. The syringealdehyde was separated by fractional recrystallization from benzene until its mass spectrum, compared with that of a mixture of syringaldehyde and vanillin in known proportions, showed it to be at least 95% pure.
At first syringaldehyde was methylated with dimethyl sulphate for one hour at 0-50°C, giving yields of 42%. It was found that heating at 70°C for a further hour increased the yield of 3,4,5-trimethoxybenzaldehyde to 56%. Heating above these temperatures lowered the yield, probably because of a Canizzaro reaction. 3,4,5-trimethoxybenzaldehyde cyanohydrin was prepared from 3,4,5-trimethoxybenzaldehyde according to the method of Kindler and Peschke4 using the bisulphite compound as an intermediate. This method eliminates the use of gaseous hydrogen cyanide and is considerably safer. 3,4,5-trimethoxybenzaldehyde cyanohydrin acetate was prepared by refluxing the cyanohydrin with acetic anhydride.
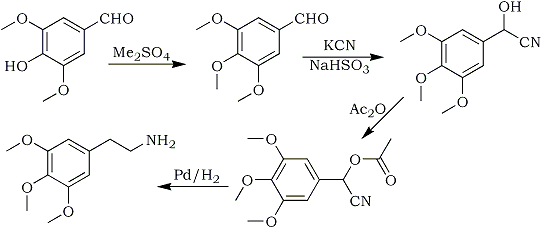
It has been reported4 that if the cyanohydrin acetate could be reduced catalytically in glacial acetic acid using palladium black as the catalyst. However, when this system was used, no mescaline was formed. Examination of the reaction mixture with a mass spectrometer indicated that the active palladium black had removed boda acetyl and cyanide groups from the 3,4,5-trimethoxybenzaldehyde cyanohydrin acetate. The reduction was successful using a less active palladium black and absolute ethanol as solvent. Mescaline was finally isolated as the sulphate.
Based on the weight of kiln-dried wood, the overall yields were 1% for E. Tegnans and 0.7% for the other two species. As the synthetic route is comparatively simple, it would appear that eucalypt sawdust can be used for the economic production of mescaline on a large scale.
Experimental
3,5-Dimethoxy-4-hydroxybenzaldehyde
Air-dry sawdust (75 g) of the species tinder investigation, nitrobenzene (45 ml) and sodium hydroxide solution (d 1.51, 2N) were added to an autoclave of 2 L capacity. The bomb was sealed, shaken vigorously for a few seconds and then agitated for 3 hours at 150°C. After cooling, the reaction mixture from the oxidation was filtered and the residue washed twice with distilled water (100 ml). The filtrate and washings were extracted with benzene to remove nitrobenzene and its reduction products; the aqueous solution was made acid with a 10% excess of concentrated hydrochloric acid and was allowed to stand for 24 hours. It was then filtered to remove the precipitate which had formed.
The clear filtrate was extracted with benzene, and the combined benzene washings were concentrated to a convenient volume (50 ml). This concentrate was extracted with sodium bisulphite solution (20%) until no more aldehyde could be detected in the washings by acidifying, boiling off the sulphur dioxide, and testing with 2,4-dinitrophenylhydrazine in hydrochloric acid (2N). The combined bisulphite washings were acidified with conc. hydrochloric acid and the sulphur dioxide removed by heating in a current of nitrogen; they were then extracted with benzene until free from aldehyde. The benzene extracts were evaporated to dryness, and then recrystallised from hot benzene. A second recrystallisation from hot water gave syringaldehyde; m.p. 111°C.
3,4,5-Trimethoxybenzaldehyde
Syringaldehyde (10 g) was dissolved with gentle heating in a solution of sodium hydroxide (3 g) in water (100 ml), and the mixture was then cooled below 50°C in an ice-bath. Dimethyl sulphate (6 ml) was added dropwise with vigorous mechanical stirring during one hour, the temperature being kept below 50°C. The mixture was then heated for one hour in a water bath at 70°C, and was cooled and extracted twice with benzene (100 ml). The benzene was evaporated and the residue was extracted with sodium bisulphite solution (20%) . After filtration, the aqueous extract was acidified and the sulphur dioxide was removed by warming in a current of nitrogen. The aldehyde was extracted with benzene and recrystallised from aqueous ethanol. Yield, 6.0 g (56%); mp 70-71°C.
3,4,5-Trimethoxybenzaldehyde Cyanohydrin
Trimethoxybenzaldehyde (8.8 g) was dissolved with gentle warming in saturated sodium bisulphite solution (20 ml). The resulting mixture was allowed to reach room temperature, when the precipitated bisulphite compound was removed by filtration and washed with absolute ethanol. The bisulphite compound was dissolved in water (10 ml) and a solution of potassium cyanide (6 g) in water (10 ml) was gradually added. The resulting oil solidified on cooling, was collected by filtration, washed first with bisulphite solution, then with water, and was finally dried over phosphorus pentoxide. Yield, 9.7g (96%); mp 81-82°C.
3,4,5-Trimethoxybenzaldehyde Cyanohydrin Acetate
3,4,5-Trimethoxybenzaldehyde cyanohydrin (10 g) was refluxed with acetic anhydride (50 ml) for 2 hours. Excess anhydride was removed by distillation under reduced pressure; the residue was taken up in ether and the ether solution was washed with sodium carbonate solution (10%), sodium bisulphite solution (20%) and water. The solution was then dried over anhydrous potassium carbonate and the residue from the dried ether distilled at 165-170°C (0.1 mm). Yield, 8.2 g (69%).
3,4,5-Trimethoxyphenethylamine
3,4,5-Trimethoxybenzaldehyde cyanohydrin acetate (1 g) was dissolved in ethanol (15 ml) and concentrated sulphuric acid (0.4 ml) and palladium black (140 mg) were added. The reduction was performed at room temperature and atmospheric pressure, and was discontinued after 95% of the calculated amount of hydrogen had been absorbed. The ethanol was eliminated and the residue was taken up in water. After filtration and extraction with ether, the aqueous solution was evaporated. The mescaline was isolated as the sulphate and was recrystallised from water. Yield, 0.5 g (50%), mp 181-184°C; lit. 183-186°C6.