Abstract
Methylenation of catechols with methylene halides was found to be catalyzed more effectively by cupric oxide in dimethylformamide.
Preparations of 1,3-benzodioxoles (methylenedioxybenzene derivatives) by methylenation of catechols have been carried out in aqueous alkaline alcoholic solvent with methylene halide or methylene sulfate as reagent, and copper powder has been employed as catalyst in some reactions3,4 However, the yield of product was usually poor, and the reaction required to be undertaken in sealed tube under nitrogen atmosphere to avoid a ready oxidation of catechols. Campbell, et al.4 reported an efficiency of bronze catalyst consisted mainly of copper, zinc, and aluminum (Tobin bronze) in the above mentioned reactions.
This paper concerns with the cupric oxide-catalyzed methylenation of catechols with methylene halides in dimethylformamide (DMF), by which 1,3-benzodioxoles are afforded in improved yield in a short reaction time.
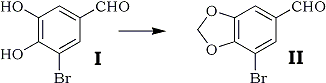
Methylenation of 3-bromo-4,5-dihydroxybenzaldehyde (I) with methylene sulfate was reported by Erne and Ramirez5. The product, 5-bromopiperonal (II), was obtained in total yield of 30% with repetition of the reaction using the recovered starting material.
Table I
Methylenation of 5-Bromoprotocatechuic Aldehyde (I)
I |
Reagent |
Catalyst |
Solvent |
K2CO3 | Bath temp (°C) |
Reaction time |
Yield of II |
10.0g |
CH2Br2 10.0 g |
CuO 1.0 g |
DMF 100 mL |
20 g | 130-140°C |
4 h |
40% |
10.0g |
CH2Br2 10.0 g |
Cu 1.0 g |
DMF 100 mL |
20 g | 130-140°C |
4 h |
27% |
10.0g |
CH2Br2 10.0 g | none |
DMF 100 mL |
20 g | 130-140°C |
4 h |
16% |
5.0g |
CH2Br2 5.0 g |
CuO 1.0 g | pyridine 50 mL |
10 g | (reflux) |
4 h |
13.3% |
5.0g |
CH2Br2 5.0 g |
Cu 1.0 g | pyridine 50 mL |
10 g | (reflux) |
4 h |
9.9% |
50.0g |
CH2Br2 50.0 g |
CuO 5.0 g |
DMF 500 mL |
100 g | 130-140°C |
4 h |
42% |
10.0g |
CH2I2 20.0 g |
CuO 1.0 g |
DMF 60 mL |
20 g | 130-140°C |
4 h |
64% |
10.0g |
CH2I2 20.0 g |
Cu2O 1.0 g |
DMF 60 mL |
20 g | 130-140°C |
4 h |
12% |
5.0g |
CH2Br2 5.0 g |
CuO 0.5 g |
DMSO 50 mL |
10 g | 130-140°C |
4 h | poor |
5.0g |
CH2Br2 5.0 g |
CuO 0.5 g |
acetone 50 mL |
10 g | (reflux) |
4 h | poor |
5.0g |
CH2Br2 5.0 g |
CuO 0.5 g |
MeOH 50 mL |
10 g | (reflux) |
4 h | poor |
5.0g |
CH2Br2 5.0 g |
Cu 0.5 g |
MeOH 50 mL |
10 g | (reflux) |
4 h | poor |
The present authors examined the above reaction in various conditions, and found that the reaction proceeded most smoothly in the presence of cupric oxide in dimethylformamide. A brief summary of the many runs is given in Table I.
The product, 5-bromopiperonal, was used as a starting material for the synthetic work of dl-cepharanthine by the authors6.
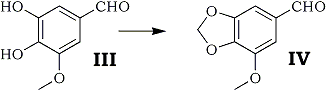
It was reported by Campbell4 that 3-methoxy-4,5-methylenedioxybenzaldehyde (myristicinaldehyde) (IV) was obtained in 35% yield by methylenation of the corresponding catechol (III) catalyzed by Tobin bronze using methylene bromide as reagent. In the same reaction catalyzed by cupric oxide in DMF, the yield of myristicin aldehyde was improved up to 72%.
On the one hand, in the reaction using metallic copper (Gattermann's copper powder) as catalyst, the yield of the product was 34%. Conditions and results are shown in Table II.
Table II.
Methylenation of 3,4-Dihydroxy-3-methoxybenzaldehyde (III)
II |
Reagent |
Catalyst |
Solvent |
K2CO3 |
Bath temp |
Reaction time |
Yield of IV |
2.0g |
CH2Br2 2.5g |
CuO 250 mg |
DMF 25 mL |
3.5 g |
135-115°C |
4 h |
72% |
2.0g |
CH2Br2 2.5g |
Cu 250 mg |
DMF 25 mL |
3.5 g |
133-145°C |
4 h |
34% |
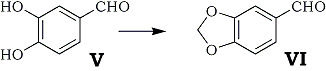
In methylenation reaction of protocatechuic aldehyde (V) catalyzed by both cupric oxide and copper powder, the yield of piperonal (VI) was 68 and 54%, respectively. The reaction conditions are summarized in Table III.
Table III.
Methylenation of Protocatechuic Aldehyde (V)
| V | CH2Br2 | Catalyst | DMF | K2CO3 | Bath temp | Reaction time | Atmosphere | Yield of VI |
| 5.0 g | 10.0 g | CuO 300 mg | 70 mL | 20 g | 100-110°C | 3 h | N2 | 68% |
| 5.0 g | 10.0 g | Cu 300 mg | 70 mL | 20 g | 100-110°C | 3 h | N2 | 51% |
Although 3,4-dihydroxy-5-methoxybenzaldehyde (III) and pyrogallol 1-methyl ether7 were shown to be readily methylenated into corresponding 1,3-benzodioxoles, any successful example of methylenation of pyrogallol has not yet been reported. From this point of view, it appeared of interest to investigate the product of the same reaction using cupric oxide as catalyst in DMF.
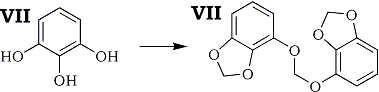
A neutral crystalline product which showed positive Labat test was obtained in 41% yield by the reaction, though any phenolic product was not isolated.
Characterization of this product was effected by NMR spectrometry and elemental analysis. These data were in accord with the structure VIII.
An attempted cleaving of methylenedioxy bridge between two 1,3-benzodioxole nuclei with methanolic hydrochloric acid was unsuccessful. The reaction conditions are given in Table IV.
Table IV.
Methylenation of Pyrogallol (VII)
VII | CH2I2 | Catalyst | DMF | K2CO3 |
Bath temp |
Reaction time |
Atmosphere |
Yield of VII |
3.0 g |
10.0 g |
CuO 250 mg |
30 mL |
20 g |
100-110°C |
3 h |
N2 |
41% |

From the data stated above, it might be concluded that methylenation of catechols with methylene halides is more effectively catalyzed by cupric oxide, and that the reaction proceeds smoothly in dimethylformamide.
However, in case of the methylenation of simple pyrocatechol, the yield of 1,3-benzodioxole was not improved by the use of catalysts involving cupric oxide or copper powder. The reaction conditions are summarized in Table V.
Table V.
Methylenation of Pyrocatechol
IX |
CH2Br2 |
Catalyst |
DMF | K2CO3 |
Bath temp |
Reaction time |
Atmosphere |
Yield of X |
10.0 g |
20.0 g |
CuO 1.0 g |
100 mL |
25 g |
100-110°C |
3 h |
N2 |
52% |
10.0 g |
20.0 g |
Cu 1.0 g |
100 mL |
25 g |
100-110°C |
3 h |
N2 |
50% |
10.0 g |
20.0 g |
none |
100 mL |
25 g |
100-110°C |
3 h |
N2 |
51% |
Experimental
General Procedure of the Reactions
Catechols, methylene halides, potassium carbonate, and catalyst were added into solvent; then the mixture was heated on an oil bath with stirring. After the reaction ended, the product was isolated and characterized as stated below. Cupric oxide catalyst was prepared from CuSO4 and NaOH2 and copper powder from CuSO4 and Zn powder.
3-Bromo-4,5-methylenedioxybenzaldehyde (5-Bromopiperonal) (II)
The reaction mixture was poured into water, and the product was extracted with ether. The ethereal extract was washed successively with 2% HCl, 2% NaOH, and water. Evaporation of the solvent after drying over anhyd. K2CO3 gave light-brown crystalline mass, which, on recrystallization from MeOH, afforded pure 5-bromopiperonal (II). Colorless pillars, mp 121-123°C (lit.5 mp 124-125°C).
3-Methoxy-4,5-methylenedioxybenzaldehyde (Myristicinaldehyde) (IV)
Recrystallization from MeOH of the crude product obtained by the same work-up method as that in 5-bromopiperonal (II) gave pure myristicinaldehyde (IV). Colorless pillars, mp 129-130°C (lit.3) mp 130-131°C).
3,4-Methylenedioxybenzaldehyde (Piperonal) (VI)
The crude product obtained by the same treatment as above was recrystallized from hexane-ether after decolorizing charcoal treatment in MeOH. Pale-yellow pillars, mp 34-34.5°C (lit.1) mp 37°C).
Methylenation of pyrogallol (VII)
Recrystallization from MeOH of the crude product obtained by the same way as above gave the pure product. Colorless pillars, mp 118-120°C. Labat test: Positive. Product identified by NMR to be bis(2,3-methylenedioxyphenoxy)methane (VIII)
1,3-Benzodioxole (X)
The reaction mixture was poured into water (500 ml); then the product was distilled with steam. The distillate was extracted with ether, and the ethereal layer was washed successively with 2% HCl, 2% NaOH, and water. Drying over anhyd. K2CO3 and evaporation of the solvent gave colorless liquid. Beilstein test: Negative.