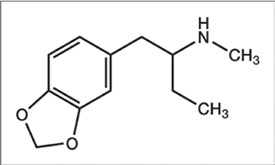
#76. MBDB
|
1-(Benzo[d][1,3]dioxol-5-yl)-N-methylbutan-2-amine
(±)-N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine
2-Methylamino-1-(3,4-methylenedioxyphenyl)butane
a-Ethyl-N-methyl-1,3-benzodioxole-5-ethanamine
N-Methyl-1-(1,3-benzodioxol-5-yl)-2-aminobutane
N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butylamine
N-Methyl-1-(3,4-methylenedioxyphenyl)-2-butanamine
EDEN
MB
MDBD
Methyl-J
(±)-N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine
2-Methylamino-1-(3,4-methylenedioxyphenyl)butane
a-Ethyl-N-methyl-1,3-benzodioxole-5-ethanamine
N-Methyl-1-(1,3-benzodioxol-5-yl)-2-aminobutane
N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butylamine
N-Methyl-1-(3,4-methylenedioxyphenyl)-2-butanamine
EDEN
MB
MDBD
Methyl-J
Registry Numbers
| CAS# | |||
| HCl salt | [128767-12-4] | ||
| Freebase | [135795-90-3] | CAS# | |
| R-(-)-Isomer HCl salt | [103882-49-1] | R-(-)-Isomer freebase | [103882-53-7] |
| S-(+)-Isomer HCl salt | [103882-50-4] | S-(+)-Isomer freebase | [103882-54-8] |
Synthesis and Chemistry
From piperonal (with Mg turnings, 1-bromopropane, diethyl ether) to 1-(1,3-benzodioxol-5-yl)butan-1-ol; (with KHSO4, distillation) to 1-(1,3-benzodioxol-5-yl)butene; (with H2O2, HCO2H) to 1-(1,3-benzodioxol-5-yl)butan-2-one; (with Al foil, HgCl2, CH3NH2Cl) to (±)-N-methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine (Nichols et al., 1986a).| C12H17NO2 | Exact Mass: 207.1259 |
| Molecular Weight: 207.27 | |
| m/z: 207.1259 (100.0%), 208.1293 (13.0%) | |
| Elemental Analysis: C, 69.54; H, 8.27; N, 6.76; O, 15.44 |
| HCl salt | m.p. 156 °C | (Nichols et al., 1986b) (IPA) |
| R-(-)-Isomer HCl salt | m.p. 192-193 °C [α]20D-26.04° | (Nichols et al., 1986b) (EtOH/Et2O) |
| S-(+)-Isomer HCl salt | m.p. 192-193 °C [α]20D+25.89° | (Nichols et al., 1986b) (EtOH/Et2O) |
Several α-ethyl phenethylamines were synthesized and analytically compared with their amphetamine counterparts (Clark et al., 1995). MS analysis of the three isomeric compounds MBDB, MDE, and MDDMA, was reported (Clark et al., 1996). N-Methyl-[11C]-labeled MBDB was synthesized and used to determine the blood-to-brain uptake in rats (Solbach et al., 1997b). Eight structurally related methylenedioxy compounds were distinguished by reversed-phase liquid chromatography and MS of the pentafluoropropionamide derivatives (DeRuiter et al., 1998b). Automated online dialysis and liquid chromatography reported MBDB in plasma and serum samples with detection limits of 10 ng/ml (Sadeghipour and Veuthey, 1998). MBDB also could be identified by FTIR spectral analysis (Praisler et al., 2000), and analyzed without derivatization by HPLC/MS (Bogusz et al., 2000). 2,3- and 3,4-isomers were synthesized and distinguished by GC-MS-MS (Borth et al., 2000). Chromatographic and spectroscopic identifications were described (Aalberg et al., 2003). MBDB was among some fifty-five phenethylamines whose fragmentation patterns were determined by a variety of mass spectrometry techniques (Kölliker and Oehme, 2004).
MDE, MDDMA, MBDB, and their 2,3-methylenedioxy positional isomers 2,3-MDE, 2,3-MDDMA, and 2,3-MBDB, all of identical mass and extremely similar mass spectra, were synthesized, derivatized, and clearly defined by GC/MS (Thigpen et al., 2007). MBDB was identified and distinguished from several structurally related compounds in human urine by capillary electrophoresis and fluorescence spectroscopy (Chung et al., 2001). An enantiomeric analysis of MBDB and its metabolite BDB was developed for rat urine (Nagai et al., 2002). A GC/MS screening of a urine sample to differentiate between MDMA, MDA, MDE, and MBDB following extraction, deconjugation, and derivatization was reported (Pellegrini et al., 2002). Analytical procedures were developed for MBDB analysis of hair samples (Junker et al., 2001; Van den Berg et al., 2002; Musshoff et al., 2002). A plasma screening and quantitative GC/MS analysis has been reported (Peters et al., 2003). Human urine was analyzed by LC-MS-MS methods (Nordgren and Beck, 2004; Nordgren et al., 2005), and serum was analyzed by extraction, derivatization with trifluoroacetic anhydride, and GC/MS (Hidvegi et al., 2006). Determination of MDMA, MDA, MDE, and MBDB in oral fluid was achieved via high performance liquid chromatography with native fluorescence detection (Concheiro et al., 2005).
Distinction of MBDB from structurally related compounds was achieved by GC/MS (Clark et al., 1996a). The nearly identical isomers, MDE and MBDB, were distinguished from one another by MS (Garofano et al., 1998), and GC/MS, with appropriate derivatization (Maruyama et al., 1998). Synthesis and spectral characterization of MBDB, BDB, GEA, and N-Me-α-Et-GEA was reported (Kanamori et al., 1999). Chromatographic and GC/MS techniques were developed for distinguishing the three isomers MDDMA, MDE, and MBDB (Aalberg et al., 2003).
Neural network analysis (NN) and neural networks coupled with principal component analysis (PC-NN) were applied to infrared spectra of stimulant and psychedelic amphetamines (Gosav et al., 2005).
Homologues and Analogues
| 3- | 4- | α- | N- | Name | CAS# | Ref |
|---|---|---|---|---|---|---|
| -OCO- | Et | Me | MBDB | (this entry) | ||
| -OCF2O- | Et | Me | F2-MBDB | [914800-83-2] | (1) | |
(1) Synthesis (Trachsel et al., 2006).
Biochemistry
With MBDB treatment, [3H]-labeled serotonin was released from rat hippocampal slices and [3H]-labeled dopamine, similarly, from rat caudate nucleus slices (Johnson et al., 1986). Stereochemical aspects of 3,4-methylenedioxymethamphetamine (MDMA) and related amphetamine derivatives' inhibition of uptake of [3H]-monoamines into synaptosomes from different regions of rat brain were evaluated (Steele et al., 1987). With S-MBDB, telemetric recordings of field potentials from frontal cortex, hippocampus, striatum, and reticular formation were made on freely moving rats (Dimpfel et al., 1989). The optical isomers of PMMA, DOM, MBDB, MDMA, and DMA were compared as stimulants in rats (Rangisetty et al., 2001). GC/MS analysis of human urine has shown that the methylenedioxy ring of MBDB opens to give DH-α-Et-MA and MH-α-Et-MA as metabolites (Maurer, 1996).
MBDB, administered orally to male rats at 20 mg/kg, was excreted in part unchanged, and as the three metabolites BDB, GEA, and N-Me-GEA (Kanamori et al., 1998a); (Nagai et al., 2002) also found MBDB and its metabolite BDB in rat urine following oral administration. Using GC/MS and LC/MS, two major routes to the observed human urinary metabolites of MBDB were N-dealkylation and demethyleneation to the catechol, and demethylation and also N-demethylation by CYP1A2 (Maurer et al., 2000). The conversion of MBDB to BDB was confirmed in human trials monitoring urine, saliva, and sweat with GC/MS; in all the biological specimens tested, MBDB was present in higher concentrations than its metabolite (Kintz, 1997). Studies were reviewed on screening procedures for detection of MDA, MDMA, MDE, BDB, and MBDB, and on their metabolism, including cytochrome P450 isoenzyme dependencies (Maurer and Kraemer, 2002).
A reproducible, simple, and small-scale method was developed for detecting the uptake and release of monoamines (dopamine, serotonin, and norepinephrine) by rat brain synaptosomes (Nagai et al., 2007).
Pharmacology
3,4-MMA and MMAI were tested for stimulus generalization in two-lever discrimination tests with rats trained with MDMA or MBDB (Johnson et al., 1991b). BDB, MBDB, and MMBDB responses were compared to one another in the newly hatched chicken, and produced behaviors more characteristic of psychedelics than amphetamine (Bronson et al., 1995a); the effects of 2C-B, MDA, MBDB, and cathinone were similarly compared (Bronson et al., 1995b). MTA, MBDB, and MMAI were compared as serotonin releasers in rats (Li et al., 1996). Biochemical and behavioral studies with MDMA and MBDB gave results similar to one another, but distinct from stimulants (amphetamine) or psychedelics (DOM, LSD); this prompted the creation of a new class name, "entactogens" (Nichols, 1986b). MBDB partially generalized for LSD in rat discrimination studies (Nichols et al., 1986a), and produced MDMA-like behaviors (Oberlender and Nichols, 1990); rats trained to discriminate between MBDB and saline, substituted MDA, MDMA, MDAI, and MDMAI completely, but not mescaline, DOM, or LSD. The term "entactogen" was further discussed for those compounds that substituted completely (Oberlender and Nichols, 1990). In establishing the definitions of entactogen, hallucinogen, and stimulant, drug discrimination-trained rats (MDMA versus (+)-amphetamine), had been used to evaluate the optical isomers of MDA, MDMA, MBDB, amphetamine, LSD, and DOM (Oberlender and Nichols, 1988). Possible neurotoxic effects of MBDB in rats were evaluated (Johnson and Nichols, 1989).
Ten suspected drug offenders (in Sweden) showed the presence of MBDB in urine at levels ranging from 0.10 to 24 µg/ml. All specimens also contained the metabolite BDB, with qualitative and quantitative GC/MS analysis of trifluoroacetic anhydride derivatives of MBDB, BDB, MDMA, MDA, and MDE (Kronstrand, 1996). Several homologues of MDA appeared in the illicit drug market in Italy (Furnari et al., 1998). The appearance of MBDB in France required careful analysis to distinguish it from its illegal, structurally close analogues and isomers (Baudot et al., 1999). GC/FTIR analysis showed that 2C-B, MDMMA, and MBDB had appeared for sale in Belgium (Dirinck et al., 2000). Employing HPLC-PDA, HPLC-MS, TLC, and NMR, a library of 35 illegal drugs was assembled and used to identify street samples (Nakashima et al., 2005). Analysis by GC/MS and LC-ESI-MS also found MBDB to be present in street drugs in Japan (Kikura-Hanajiri et al., 2005). In Japan, homo-MDMA was sold on the street under the recognized name MBDB (Matsumoto et al., 2006). Syntheses and analytical methods for MBDB and its analogues, including BDB, MDA, MDE, MDMA, and methylone, were developed to support forensic investigations (Sanuki et al., 2006). A thorough review article highlighted the distinction of MBDB (analytically and pharmacologically) from its close analogues and homologues (Van Aerts et al., 2000).
Two deaths were established as having been caused by MBDB (Carter et al., 2000).
MBDB's S-(+)-isomer is orally active in humans at 125 mg, the racemate is active at 150-210 mg (Nichols et al., 1986a); the racemate was active at 180-210 mg; duration 4-6 hours (Shulgin and Shulgin, 1991).
Legal Status
MBDB is not a scheduled compound under federal drug law, or under District of Columbia or any state laws. MBDB has been regulated in Japan by their Narcotics and Psychotropics Control Law since April 22, 2006 (Sanuki et al., 2006).